I have always been interested in the science of cooking. Most times when we cook we add ingredients to the recipe not knowing why it is done. Asking ‘WHY’ something happens when you add an ingredient and understanding the science may or may not improve your cooking but for me it is a learning experience. I had completed a Coursera course called ‘Science of Gastronomy’ through Hong Kong University which was a free course and very interesting. There is an MIT EDX course on Food Fermentation- The Science of Cooking going on right now which is discussing the roles of microbes in food. If you are interested also get the book series- What Einstein Told His Cook- Kitchen Science Explained. I am also now reading Masala Lab – The Science of Indian Cooking by Krish Ashok. These tips are either from these books or information I learned over time from other books, the internet, or discussing this with my scientist husband. I am hoping to write more on this over the year and as I learn more.
I learned about pH scale in high school and college and I don’t think we were taught real world applications. As a parent, who always tried to answer every ‘why’, this was pre-google days and later as a teacher always encouraging students to ask questions and finding out if I do not know the answer has made me also wonder and learn. Teaching pH to my 5th graders made me learn more about it too. Knowing this, helps me problem solve if my food is too salty or sour.
Today pH is also spoken a lot about in the new alkaline diet. Ayurveda has been talking about this for a long time. Outside the kitchen, gardeners are interested in the pH of the soil which is very important for certain plants.
If you want a fun activity, to make pH indicators, with young children, here is a link using red cabbage. We did this at school and the kids loved it. We used spray bottles with soap and other ingredients and made tye and dye.
https://science.discoveryplace.org/stay-at-home-science/kitchen-chemistry
https://www.steampoweredfamily.com/activities/acids-bases-ph-chemistry-experiment/
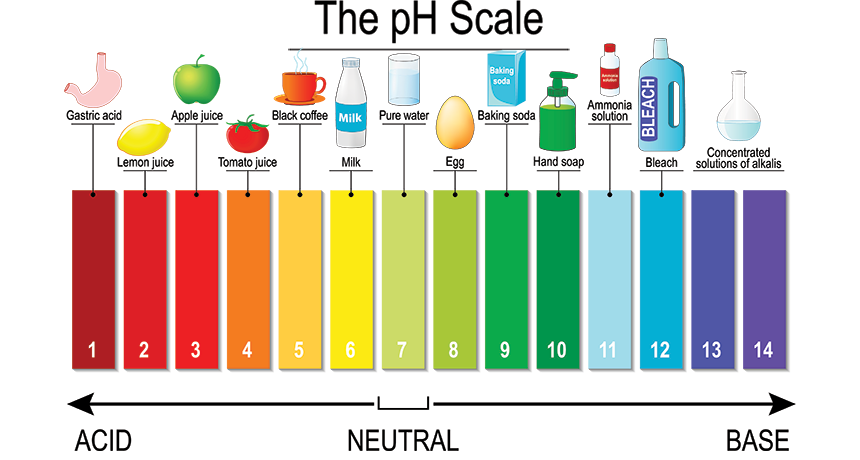

The image above is a pH scale that explains which food is acidic, neutral or basic. pH is a quantitative measurement of the acidity or basicity of liquids. This is a basic definition of pH. If you want more information, here is Khan academy explaining pH https://www.khanacademy.org/science/biology/water-acids-and-bases/acids-bases-and-ph/a/acids-bases-ph-and-bufffers
Some ingredients which are not part of the table:
- vinegar pH 2.0
- honey pH 3.9 (did not realize that)
- wine pH 4.0 – Wine will fall between vinegar and tomatoes in acidity
- buttermilk and yogurt pH 4.4
- cheese pH 4.4 to 6.8
- natural cocoa pH 5.6
So why do we need to know about pH when we cook? Most of us have learned from experience, parents, and maybe our grandmothers, to add something to neutralize something else. For example, if something is salty, you would add more water or add a potato to absorb the salt. If you are making tomato sauce or rasam and have added too much vinegar or tamarind and need to reduce the acid, we know we could dilute it with water or add something spicy to it. Would you add spices like chilies to it? You could add cream or coconut milk to reduce the acidity but what if you did not want the creamy element to the dish? Why would you add cream? The fat in the cream cuts the acidity.
I was recently making tomato sauce and it was very acidic. I learned from a discussion my son, husband and I were having as to what to add to reduce the acidity. It was a long discussion on acids and bases and what could be added to neutralize the acid. We decided that adding baking soda helps if food is very acidic. I had never thought of adding baking soda as baking soda usually has a bad rap. Lo and behold, added a pinch of soda and it really helped. Why did it help? Knowing the science behind it helps. If you look at the pH scale, you will see that tomatoes are acidic. So, to reduce the acid, you will have to add something basic, which is baking soda.
When we cook dried garbanzo beans (chickpeas), we usually soak the beans for 8 hours. We have been told to add a pinch of baking soda to the water when cooking the beans and from experience, we know that it cooks faster. Do you know why? My friend in college who is from Punjab introduced me to the idea of adding a teabag to the beans while cooking. It gives a beautiful dark brown color and I thought that was the reason until I read Masala Lab. In the book, Krish Ashok explains that by adding baking soda, the soda reacts with the pectin in the skin of the beans and breaks it down faster, and cooks faster. Tea is slightly acidic and soda is basic, so the tea (acid) also neutralizes all the unused baking soda (base). Isn’t that interesting?
Here are some ways to use acid to make food taste/look better:
- Enhance your dishes with acid. A dash of lemon juice or vinegar always perks up a dish. Adding some balsamic vinegar to tomato sauce brings it to a different level
- Adding lemon juice on cut avocado, banana or cut apples reduce oxidation (by reducing the pH on the cut surface and slowing down the chemical reaction) and avoid discoloration
- Adding cream, cheese (dairy) which is slightly acidic adds a rich flavor
- Adding some yogurt in some water while cutting vegetable and storing till cooking time like potatoes and raw plantain reduces discoloration
- Fermenting food increases the acidity in the fermented food. Why? Fermentation is a natural process through which beneficial microorganisms like yeast and bacteria convert carbohydrates — such as starch and sugar — into alcohol or acids making food more nutritious and preserving it so people can store it for longer periods of time without it spoiling.
- A pinch of sugar reduces the acidity of the food
- A pinch (or more depending on the acidity) of baking soda reduces acidity in the food
Do you have more tips to add. Please do let me know by commenting. Please share for more ‘why’ questions.

Interesting
LikeLike
Thank you, Sowmya.
LikeLiked by 1 person
Hi Preethi! Love learning about the chemistry of cooking! Thanks for writing this! Myrna Sent from my iPhone
>
LikeLike
Thank you, Myrna! 🙂
LikeLike
Ah food science. It’s all part of the fun of cooking. Great article, really informative and love all the pictures.
LikeLike
Thank you so much! Glad you enjoyed the post.
LikeLike
wow this quite a lesson. well done.
LikeLike
Thank you, Sophia! Glad you enjoyed it.
LikeLiked by 1 person